REGIONAL SETTING
(from Pape, 2013)
 Photo 1. Semidesert grassland habitat at Kartchner Caverns State Park.
Photo 1. Semidesert grassland habitat at Kartchner Caverns State Park.
Kartchner Caverns is located within Kartchner Caverns State Park (KCSP) near the town of Benson, Cochise County, Arizona. The Park lies within the Basin and Range Province physiographic region of the western United States. The cave is contained within a grouping of small hills situated low on the east flank of the Whetstone Mountains. The cave is formed in Escabrosa limestone of Mississippian age (Jagnow 1999), and dating of speleothems from the cave provide a minimum age for the cave of nearly 200 ka (Ford and Hill 1999). The collapse-sink entrance of the cave is located on a southeasterly-facing slope at an elevation of 1,428 m. The vegetation community in the area is dominantly the Semidesert Grassland biome (Photo 1) as described by Brown (1982).
Average annual precipitation recorded at the Park over the last 20 years is 30.2 centimeters. Less than two percent of this water potentially reaches the cave through infiltration as autogenic meteoric drip waters (Buecher 1999). Compared with earlier, more mesic (wetter) periods, the geologically recent desertification of the southwestern United States, beginning approximately 12–10 kya years ago (Van Devender 1990), resulted in less precipitation being available for the meteoric recharge and maintenance of humid cave environments. As a result, many caves in the region, particularly those with multiple entrances that permit significant air exchange with the surface, are probably considerably drier than they were prior to desertification. Caves such as Kartchner Caverns, which still support a high humidity environment, may serve as refugia for macro-invertebrates that have been removed regionally from the epigean environment as a result of the recent desertification.
Average annual precipitation recorded at the Park over the last 20 years is 30.2 centimeters. Less than two percent of this water potentially reaches the cave through infiltration as autogenic meteoric drip waters (Buecher 1999). Compared with earlier, more mesic (wetter) periods, the geologically recent desertification of the southwestern United States, beginning approximately 12–10 kya years ago (Van Devender 1990), resulted in less precipitation being available for the meteoric recharge and maintenance of humid cave environments. As a result, many caves in the region, particularly those with multiple entrances that permit significant air exchange with the surface, are probably considerably drier than they were prior to desertification. Caves such as Kartchner Caverns, which still support a high humidity environment, may serve as refugia for macro-invertebrates that have been removed regionally from the epigean environment as a result of the recent desertification.
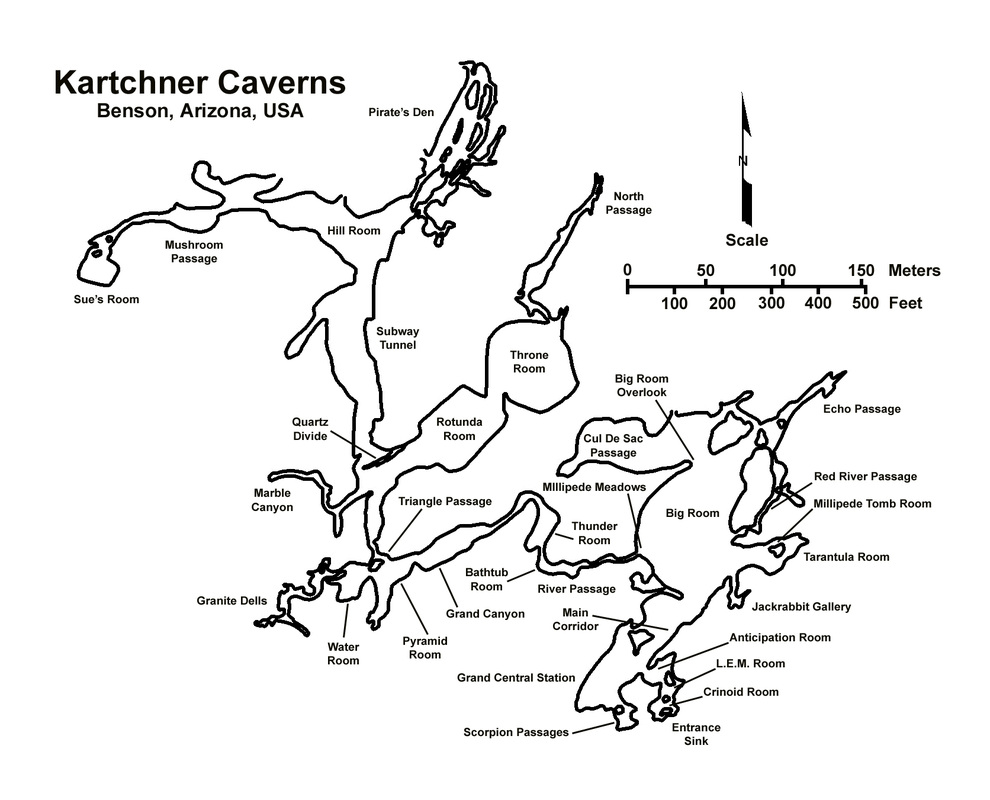
MAP OF THE CAVERNS
THE CAVE ENVIRONMENT
(from Pape and OConnor, 2014)
 Photo 2. Flood waters in the Lake Room. Note the two markers; from February 19, 1991 and 19 years later, on February 13, 2010.
Photo 2. Flood waters in the Lake Room. Note the two markers; from February 19, 1991 and 19 years later, on February 13, 2010.
Survival of cave-adapted species is dependent on maintenance of cave microclimatic parameters that are often in precarious balance with the influence of surface environmental conditions. This is particularly true in arid environments such as the southwestern U.S., where the combination of extreme summer temperatures and low humidity most of the year result in evaporation rates at the surface that greatly exceed annual precipitation. Aggravating the effects of surface conditions on the cave environment are the effects of air flow in caves. Caves with multiple entrances commonly suffer from a “chimney effect” air circulation pattern, which can result in drying of the cave, adversely affecting the habitat of resident invertebrates (and other biota). Additional pressures on regional cave ecosystems include a recent, ongoing drought (Breshears et al. 2005) and the effects of regional and global climate change.
In many respects Kartchner Caverns is typical of caves in southern Arizona. The cave does not have significant aquatic components such as a perennial stream or much in the way of other persistent waters such as perched pools. Because of this there is no obvious aquatic macroinvertebrate component of its resident biota. The cave is still active, however, with autogenic meteoric waters infiltrating the cave from above depositing calcite as speleothems, and allogenic waters periodically flooding portions of the cave during extended or stochastic precipitation events higher in the watershed. Water from these surface runoff events enters the cave from ephemeral desert streams that flank the cave hill on its west and south sides. The flooding can be extensive, and may reach depths up to 2.4 meters in some areas of the cave. Interestingly, the floodwaters last only a few weeks at most before penetrating to humanly inaccessible lower levels of the sedimentary section within which the cave is developed.
One of these major flood events occurred during the initial study, and it was observed that organic debris such as leaves and twigs had entered the cave with the floodwaters. Invertebrates, particularly the terrestrial cave isopod (Brackenridgia nr. sphinxensis), were observed feeding on these materials (Welbourn 1999). By chance, a major flood event of almost identical magnitude occurred during the recent study (Photo 2), and no associated coarse organic materials were observed in the cave. We suspect that there may have been a change in the influx hydrology of the cave such that coarse materials now get filtered out before reaching the cave interior. These flood events certainly bring some dissolved carbon and other nutrients into the cave, but these may be inadequate to support a regular presence of macro-invertebrates in these areas.
A significant component of these occasional cave flood events is suspended sediments, primarily clays, which are deposited as a thin layer over the entire substrate with each event. Due to the accumulation of these sediments there are no exposed pieces of debris on the floors that provide shelter habitat for invertebrates in areas of the cave subject to flooding. The exposed post-flood substrate consists of large-scale polygonal blocks of accumulated sediments, which typically remain saturated for long periods (Photo 3). The exposed post-flood substrate consists of large-scale polygonal blocks of accumulated sediments, which typically remain saturated for long periods (Photo 3). The exposed post-flood substrate consists of large-scale polygonal blocks of accumulated sediments, which typically remain saturated for long periods (Photo 3). The polygonal structure of the substrate indicates that these areas do occasionally dry to a point adequate for the formation of these shapes (desiccation cracks), which result from shrinkage of clays as moisture is lost from the soil. We suspect that the combination of an apparent limited nutrient supply along with the lack of shelter habitat is sufficient evidence to explain the absence of macro-invertebrates in areas of the cave subject to occasional stochastic flood events. Other unidentified factors may also be involved.
In many respects Kartchner Caverns is typical of caves in southern Arizona. The cave does not have significant aquatic components such as a perennial stream or much in the way of other persistent waters such as perched pools. Because of this there is no obvious aquatic macroinvertebrate component of its resident biota. The cave is still active, however, with autogenic meteoric waters infiltrating the cave from above depositing calcite as speleothems, and allogenic waters periodically flooding portions of the cave during extended or stochastic precipitation events higher in the watershed. Water from these surface runoff events enters the cave from ephemeral desert streams that flank the cave hill on its west and south sides. The flooding can be extensive, and may reach depths up to 2.4 meters in some areas of the cave. Interestingly, the floodwaters last only a few weeks at most before penetrating to humanly inaccessible lower levels of the sedimentary section within which the cave is developed.
One of these major flood events occurred during the initial study, and it was observed that organic debris such as leaves and twigs had entered the cave with the floodwaters. Invertebrates, particularly the terrestrial cave isopod (Brackenridgia nr. sphinxensis), were observed feeding on these materials (Welbourn 1999). By chance, a major flood event of almost identical magnitude occurred during the recent study (Photo 2), and no associated coarse organic materials were observed in the cave. We suspect that there may have been a change in the influx hydrology of the cave such that coarse materials now get filtered out before reaching the cave interior. These flood events certainly bring some dissolved carbon and other nutrients into the cave, but these may be inadequate to support a regular presence of macro-invertebrates in these areas.
A significant component of these occasional cave flood events is suspended sediments, primarily clays, which are deposited as a thin layer over the entire substrate with each event. Due to the accumulation of these sediments there are no exposed pieces of debris on the floors that provide shelter habitat for invertebrates in areas of the cave subject to flooding. The exposed post-flood substrate consists of large-scale polygonal blocks of accumulated sediments, which typically remain saturated for long periods (Photo 3). The exposed post-flood substrate consists of large-scale polygonal blocks of accumulated sediments, which typically remain saturated for long periods (Photo 3). The exposed post-flood substrate consists of large-scale polygonal blocks of accumulated sediments, which typically remain saturated for long periods (Photo 3). The polygonal structure of the substrate indicates that these areas do occasionally dry to a point adequate for the formation of these shapes (desiccation cracks), which result from shrinkage of clays as moisture is lost from the soil. We suspect that the combination of an apparent limited nutrient supply along with the lack of shelter habitat is sufficient evidence to explain the absence of macro-invertebrates in areas of the cave subject to occasional stochastic flood events. Other unidentified factors may also be involved.
|
The average air temperature in a given cave typically stabilizes at the mean annual surface temperature, which is a function of the elevation and latitude where the cave is located. Based on these criteria, the air temperature expected in Kartchner Caverns should be 15.4°C. However, while the temperature in the cave fluctuates it always remains above this level (between 18.6°C and 21.3° C). Buecher (1999) suggested that the elevated air temperature in Kartchner Caverns may be due to proximity to geothermal waters in the San Pedro Valley, where the cave is located. Other factors may be responsible or may contribute to this effect. Relative humidity in the cave varies primarily in association with proximity to surface connections. Values in the deeper reaches of the cave are relatively constant, and during the initial study averaged 99.42 percent (Buecher 1999).
|
NUTRIENT SOURCES
(from Pape and OConnor, 2014)
Compared with epigean habitats cave environments are often nutrient poor and resources that do occur are mostly transported into caves from allochthonous sources. Methods of nutrient energy transfer into caves vary, but often include infusion of organic materials transported by animals, or through physical processes such as allogenic stream flow and autogenic meteoric waters percolating through the overburden above the cave. Influx of autogenic meteoric waters, which historically was not considered an important source of nutrient transport into caves, has recently been recognized as a source of significant quantities of organics that are derived from the epikarst (Culver and Pipan 2009; Romero 2009).
Animals that use caves for a portion of their life cycle, but which feed primarily outside of caves, are an important element of organic nutrient input to cave ecosystems. Therefore, unimpeded movement of biota, including both invertebrates and vertebrates, is often critical to the maintenance of many cave food webs. A significant portion of the organic nutrients brought into Kartchner Caverns is contributed by animals. The primary source of these nutrients (by volume) is the annual influx of bat guano provided by the cave myotis maternity colony. Other animals that bring measurable amounts of nutrients into the cave in their feces include cave crickets (Ceuthophilus pima), ringtails (Bassariscus astutus), and rodents that occasionally enter the cave. Animals occurring as incidental occurrences in the cave, including invertebrates and the occasional vertebrate, which fail to find their way out of the cave, become a food source for resident species as prey or carcasses. Natural death of resident invertebrates retains nutrients that are recycled within the cave food webs.
Based on our observations we conclude that coarse organic debris, such as plant litter, is not a major nutrient resource available to cave macro-invertebrates living near cave surface connections at Kartchner Caverns. This condition is not unusual for caves in desert southwestern region of the U.S.
Autochthonous nutrients present in the cave are probably limited to microbial elements. Some of the primary consumers in the cave, particularly collembola, may use these resources to some degree. Otherwise, most macro-invertebrates in the cave probably subsist primarily on allochthonous nutrient input from the sources previously discussed.
The presence of large numbers of people moving through caves on tours of commercially-developed caves inevitably results in the cumulative deposition of lint composed of clothing fiber and human hair and skin elements. This unintended input provides nutrients for microbes and arthropods. This input is minimized at Kartchner Caverns by the curbed trails, which contain much of this litter, and the regular washing down of the trail system, which removes it.
Artificial lighting in show caves provides energy for the development of phototrophic lampenflora. Lampenflora are initially comprised of prokaryotic cyanobacteria and eukaryotic algae which form on surfaces of rock and speleothems adjacent to the lights (Mulec and Kosi 2009). Lampenflora provide a nutrient base for microbes and primary consumer level arthropods.
Animals that use caves for a portion of their life cycle, but which feed primarily outside of caves, are an important element of organic nutrient input to cave ecosystems. Therefore, unimpeded movement of biota, including both invertebrates and vertebrates, is often critical to the maintenance of many cave food webs. A significant portion of the organic nutrients brought into Kartchner Caverns is contributed by animals. The primary source of these nutrients (by volume) is the annual influx of bat guano provided by the cave myotis maternity colony. Other animals that bring measurable amounts of nutrients into the cave in their feces include cave crickets (Ceuthophilus pima), ringtails (Bassariscus astutus), and rodents that occasionally enter the cave. Animals occurring as incidental occurrences in the cave, including invertebrates and the occasional vertebrate, which fail to find their way out of the cave, become a food source for resident species as prey or carcasses. Natural death of resident invertebrates retains nutrients that are recycled within the cave food webs.
Based on our observations we conclude that coarse organic debris, such as plant litter, is not a major nutrient resource available to cave macro-invertebrates living near cave surface connections at Kartchner Caverns. This condition is not unusual for caves in desert southwestern region of the U.S.
Autochthonous nutrients present in the cave are probably limited to microbial elements. Some of the primary consumers in the cave, particularly collembola, may use these resources to some degree. Otherwise, most macro-invertebrates in the cave probably subsist primarily on allochthonous nutrient input from the sources previously discussed.
The presence of large numbers of people moving through caves on tours of commercially-developed caves inevitably results in the cumulative deposition of lint composed of clothing fiber and human hair and skin elements. This unintended input provides nutrients for microbes and arthropods. This input is minimized at Kartchner Caverns by the curbed trails, which contain much of this litter, and the regular washing down of the trail system, which removes it.
Artificial lighting in show caves provides energy for the development of phototrophic lampenflora. Lampenflora are initially comprised of prokaryotic cyanobacteria and eukaryotic algae which form on surfaces of rock and speleothems adjacent to the lights (Mulec and Kosi 2009). Lampenflora provide a nutrient base for microbes and primary consumer level arthropods.
Ecology of the Macro-invertebrates of Kartchner Caverns
(from Pape and OConnor, 2014)
 Photo 4. Plant roots hanging from the ceiling at Sharon's Saddle in the Big Room.
Photo 4. Plant roots hanging from the ceiling at Sharon's Saddle in the Big Room.
There are two major categories of habitats that may support macro-invertebrates in caves: aquatic and terrestrial. Aquatic habitats in Kartchner Caverns are very limited as there are very few perennial water sources in the cave. Temporary waters collect seasonally in very small pools in response to the influx of autogenic meteoric waters during the two regional precipitation seasons. While some of these pools may last for many months, they are not perennial. Most of the few perennial pools occurring in the cave are at the base of the Big Wall in the Throne Room. On an irregular basis when there is adequate sustained flow down the ephemeral surface washes in the Park
there can be significant flooding of portions of the cave. As mentioned previously, these waters infiltrate completely after several months, and are thus also not perennial in humanly accessible portions of the cave system.
There are almost certainly copepods (Crustacea: Copepoda) and other micro-invertebrates that live in the epikarst that are transported into the cave by the seasonal influx of autogenic meteoric waters. These animals may persist on wet flowstone and in the temporary pools in the cave as long as free water is present. These animals may be an important food source for the few macroinvertebrates that live on wet formations in the cave. The only invertebrates that we have regularly observed on wet formations are sminthurid collembolans and a small unidentified spider that spins its webs at these sites, and likely prey on the collembolans. The sminthurids have also been found on the surface of small pools of water, again possibly making use of microfauna in the water. Although all four individuals of the cambalid millipede we found in the cave were dead, they were found on wet formations. We suspect that the cambalid millipede may be part of the ecology of a wet flowstone biofilm community that is supported by autogenic meteoric waters. Although associated with wet environments, these arthropods are considered terrestrial animals.
Food webs in the cave are partitioned by a combination of various microclimatic conditions and available nutrients. The bat guano deposits are mostly a contained ecosystem, and one that is totally dependent on the annual influx of fresh nutrients brought into the cave each year by the bat colony. There are species, primarily mites, which are found only on the bat guano deposits. And, there are several invertebrates that are associated with the guano deposits, but are only present peripherally, apparently avoiding the highly competitive environment of the guano deposits.
Most of the macro-invertebrate activity in the cave is associated with areas that do not flood and that provide nutrient resources of some kind. In these higher, “drier” portions of the cave the macro-invertebrates can establish and maintain themselves undisturbed for many
generations. These areas are more stable and hospitable to cave life than the portions of the cave that are subject to occasional stochastic flooding events. These areas typically also contain an abundance of breakdown blocks and a multitude of floor debris consisting of small to large rocks and pieces of broken calcite formations that provide shelter habitat for macro-invertebrates. Some floor areas that are covered with flowstone have sub-floor areas that also provide habitat for invertebrates, such as occurs in the Jackrabbit Gallery.
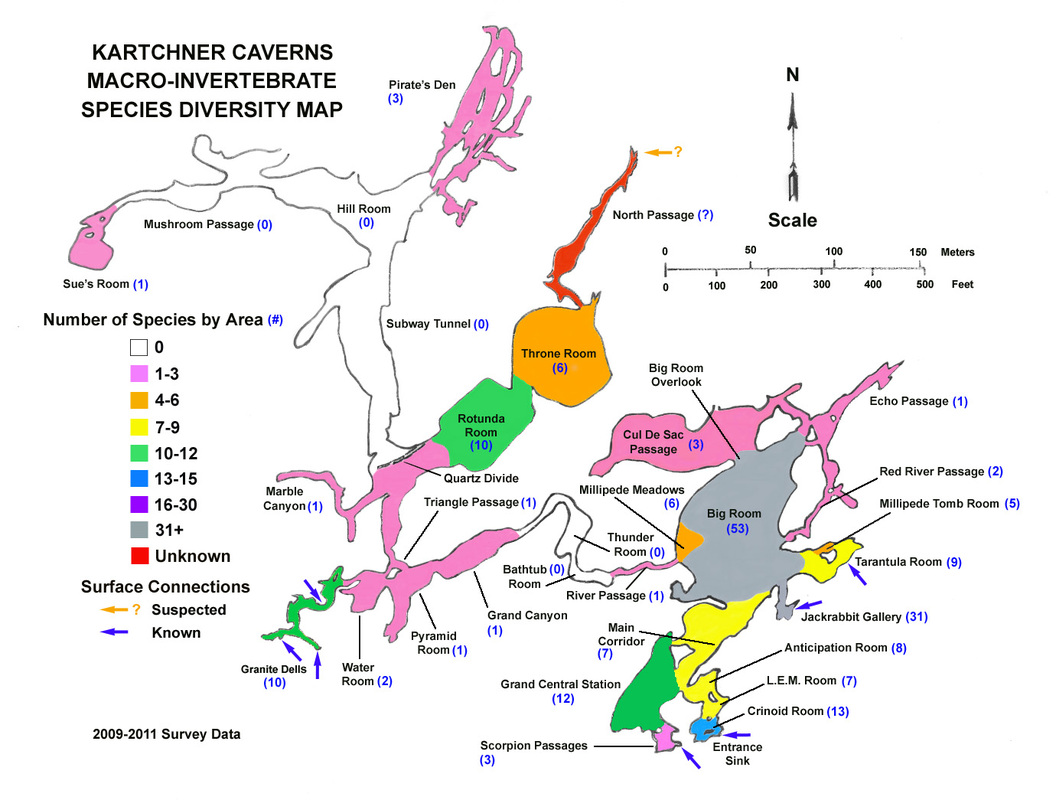
Overall, surface connection areas are some of the most ecologically significant areas of the cave, cumulatively supporting a number of non-incidental species (48) nearly equal to that of the bat guano habitats (53) in the Big Room. If one were to remove the significant mite component of 16 species directly associated with the bat guano deposits in the Big Room, the surface connection areas would then exceed the number of species supported by bat guano.
One habitat present in a few areas of the cave, but which is apparently underutilized by macro-invertebrates, is exposed plant roots that penetrate the cave and hang from walls or ceiling areas. This nutrient resource is often used by invertebrates in caves. The best example of this habitat in the cave is in the south end of the Big Room at Sharon’s Saddle (Photo 4). Here, the roots are seasonally wet and have enough biomass to theoretically support small numbers of invertebrates. Other areas where roots are exposed include the upper Tarantula Room and two of the surface connections in the Granite Dells area. Some of the roots in the upper Tarantula Room have been used as food by termites, but only where the roots are in contact with wall surfaces, which allows the termites to enclose them with their soil tunnels that they have extended from the soil horizon into the cave (Photo 5).
there can be significant flooding of portions of the cave. As mentioned previously, these waters infiltrate completely after several months, and are thus also not perennial in humanly accessible portions of the cave system.
There are almost certainly copepods (Crustacea: Copepoda) and other micro-invertebrates that live in the epikarst that are transported into the cave by the seasonal influx of autogenic meteoric waters. These animals may persist on wet flowstone and in the temporary pools in the cave as long as free water is present. These animals may be an important food source for the few macroinvertebrates that live on wet formations in the cave. The only invertebrates that we have regularly observed on wet formations are sminthurid collembolans and a small unidentified spider that spins its webs at these sites, and likely prey on the collembolans. The sminthurids have also been found on the surface of small pools of water, again possibly making use of microfauna in the water. Although all four individuals of the cambalid millipede we found in the cave were dead, they were found on wet formations. We suspect that the cambalid millipede may be part of the ecology of a wet flowstone biofilm community that is supported by autogenic meteoric waters. Although associated with wet environments, these arthropods are considered terrestrial animals.
Food webs in the cave are partitioned by a combination of various microclimatic conditions and available nutrients. The bat guano deposits are mostly a contained ecosystem, and one that is totally dependent on the annual influx of fresh nutrients brought into the cave each year by the bat colony. There are species, primarily mites, which are found only on the bat guano deposits. And, there are several invertebrates that are associated with the guano deposits, but are only present peripherally, apparently avoiding the highly competitive environment of the guano deposits.
Most of the macro-invertebrate activity in the cave is associated with areas that do not flood and that provide nutrient resources of some kind. In these higher, “drier” portions of the cave the macro-invertebrates can establish and maintain themselves undisturbed for many
generations. These areas are more stable and hospitable to cave life than the portions of the cave that are subject to occasional stochastic flooding events. These areas typically also contain an abundance of breakdown blocks and a multitude of floor debris consisting of small to large rocks and pieces of broken calcite formations that provide shelter habitat for macro-invertebrates. Some floor areas that are covered with flowstone have sub-floor areas that also provide habitat for invertebrates, such as occurs in the Jackrabbit Gallery.
Overall, surface connection areas are some of the most ecologically significant areas of the cave, cumulatively supporting a number of non-incidental species (48) nearly equal to that of the bat guano habitats (53) in the Big Room. If one were to remove the significant mite component of 16 species directly associated with the bat guano deposits in the Big Room, the surface connection areas would then exceed the number of species supported by bat guano.
One habitat present in a few areas of the cave, but which is apparently underutilized by macro-invertebrates, is exposed plant roots that penetrate the cave and hang from walls or ceiling areas. This nutrient resource is often used by invertebrates in caves. The best example of this habitat in the cave is in the south end of the Big Room at Sharon’s Saddle (Photo 4). Here, the roots are seasonally wet and have enough biomass to theoretically support small numbers of invertebrates. Other areas where roots are exposed include the upper Tarantula Room and two of the surface connections in the Granite Dells area. Some of the roots in the upper Tarantula Room have been used as food by termites, but only where the roots are in contact with wall surfaces, which allows the termites to enclose them with their soil tunnels that they have extended from the soil horizon into the cave (Photo 5).
 Photo 5. Termite mud tunnels enclosing plant root. The root was growing from the surface down into the cave, and between the bedrock wall and old flowstone surface coating the wall. Condensation corrosion dissolution of the contact between the two layers resulted in a space that was capitalized on by the root.
Photo 5. Termite mud tunnels enclosing plant root. The root was growing from the surface down into the cave, and between the bedrock wall and old flowstone surface coating the wall. Condensation corrosion dissolution of the contact between the two layers resulted in a space that was capitalized on by the root.
A lampenflora faunal community has been introduced into the cave and is artificially distributed incidental to the placement of the tour lights. Ultimately, species of mosses and ferns could potentially colonize these areas, but this level of lampenflora development is not often observed (or tolerated) in commercial caves. Because lampenflora can develop over a wide humidity range, these communities are less constrained by moisture requirements than some of the other food web habitats.
There is a suite of macro-invertebrate taxa that are commonly present in caves in southern Arizona. Peculiarly, none of the caves apparently supports all of these species, and few caves have the same mix of these species as elements of their fauna. Kartchner Caverns fits within this pattern. Taxa that are commonly represented in this core of cave residents in the region include a cave cricket (Ceuthophilus sp.), a laniatore harvestman (Sitalcina sp.), a trichoniscid isopod (Brackenridgia sp.), a cave carabid beetle (Agonum sp. [subgenus Rhadine]), and one or more small spider species (some of which are cave-adapted), usually in the families Hahniidae (e.g. Neocryphoeca sp.), Leptonetidae (e.g. Leptoneta sp.), Nesticidae (e.g. Eidmanella sp.), or Theridiidae (e.g. Achaearanea sp. and Thymoites sp.). The species in these groups present in Kartchner Caverns include Ceuthophilus pima, Brackenridgia sp., two undescribed species of Neocryphoeca, Eidmannella pallida, and Achaearanea canionis. We have found no representative species of Sitalcina, Rhadine, or Leptoneta in the cave.
As of the spring of 2016 we have documented 99 species of macro-invertebrates that have a positive association with the ecology of the cave. The figure below shows the relative distribution of species richness in the cave (through 2014). Since many species occur in multiple areas of the cave the individual numbers are not additive to the total.
There is a suite of macro-invertebrate taxa that are commonly present in caves in southern Arizona. Peculiarly, none of the caves apparently supports all of these species, and few caves have the same mix of these species as elements of their fauna. Kartchner Caverns fits within this pattern. Taxa that are commonly represented in this core of cave residents in the region include a cave cricket (Ceuthophilus sp.), a laniatore harvestman (Sitalcina sp.), a trichoniscid isopod (Brackenridgia sp.), a cave carabid beetle (Agonum sp. [subgenus Rhadine]), and one or more small spider species (some of which are cave-adapted), usually in the families Hahniidae (e.g. Neocryphoeca sp.), Leptonetidae (e.g. Leptoneta sp.), Nesticidae (e.g. Eidmanella sp.), or Theridiidae (e.g. Achaearanea sp. and Thymoites sp.). The species in these groups present in Kartchner Caverns include Ceuthophilus pima, Brackenridgia sp., two undescribed species of Neocryphoeca, Eidmannella pallida, and Achaearanea canionis. We have found no representative species of Sitalcina, Rhadine, or Leptoneta in the cave.
As of the spring of 2016 we have documented 99 species of macro-invertebrates that have a positive association with the ecology of the cave. The figure below shows the relative distribution of species richness in the cave (through 2014). Since many species occur in multiple areas of the cave the individual numbers are not additive to the total.
© 2023 R.B. Pape. All rights reserved.